Genetic Code #6 - Zwitterions Mystery Cache
Genetic Code #6 - Zwitterions
Please note Use of geocaching.com services is subject to the terms and conditions
in our disclaimer.
Cache is not at the posted coordinates! As a geo-geneticist, you must solve the puzzle below:
In chemistry, a zwitterion is a neutral molecule with a positive and a negative electrical charge, distinct from dipoles, at different locations within that molecule. Unlike simple amphoteric compounds that might only form either a cationic or anionic species depending on external conditions, a zwitterion simultaneously has both ionic states in the same molecule.
Amino acids are the best-known examples of zwitterions. The amine and carboxylic acid functional groups found in amino acids allow them to have amphiprotic properties. Carboxylic acid groups can be deprotonated to become negative carboxylates, and α-amino groups can be protonated to become positive α-ammonium groups. At pH values greater than the pKa of the carboxylic acid group (mean for the 20 common α-amino acids is about 2.2), the negative carboxylate ion predominates. At pH values lower than the pKa of the α-ammonium group (mean for the 20 common α-amino acids is about 9.4), the nitrogen is predominantly protonated as a positively charged α-ammonium group. Thus, at pH between 2.2 and 9.4, the predominant form adopted by α-amino acids contains a negative carboxylate and a positive α-ammonium group, so has net zero charge. This molecular state is known as a zwitterion, from the German Zwitter meaning hermaphrodite or hybrid.
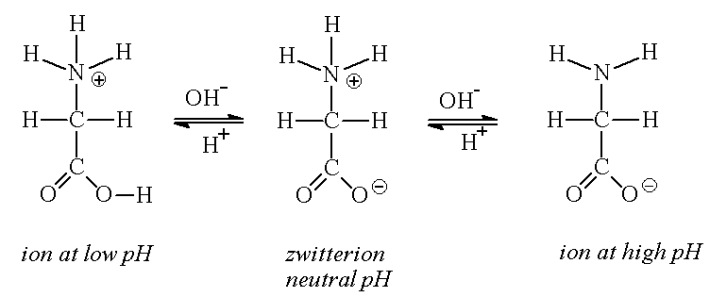 At pH values between the two pKa values, the zwitterion predominates, but coexists in dynamic equilibrium with small amounts of net negative and net positive ions. At the exact midpoint between the two pKa values, the trace amount of net negative and trace of net positive ions exactly balance, so that average net charge of all forms present is zero. This pH is known as the isoelectric point pI, so pI = ½(pKa1 + pKa2). Each individual amino acid has a unique isoelectric point. Zwitterions have minimum solubility at their isolectric point and some amino acids can be isolated by precipitation from water by adjusting the pH to the required isoelectric point.
At pH values between the two pKa values, the zwitterion predominates, but coexists in dynamic equilibrium with small amounts of net negative and net positive ions. At the exact midpoint between the two pKa values, the trace amount of net negative and trace of net positive ions exactly balance, so that average net charge of all forms present is zero. This pH is known as the isoelectric point pI, so pI = ½(pKa1 + pKa2). Each individual amino acid has a unique isoelectric point. Zwitterions have minimum solubility at their isolectric point and some amino acids can be isolated by precipitation from water by adjusting the pH to the required isoelectric point.
For more info see Wikipedia: Amino Acids
Puzzle:
N 32° 4-Arg.His-Lys-Tyr W 097° Asp-8.Arg-Phe-Arg
***Do not forget the Bonus Clue.***

You can validate your puzzle solution with certitude.
Additional Hints
(Decrypt)
Chmmyr: sbe ahzoref terngre 10, hfr gur barf cynpr
Pnpur: qrpba va gerr
Treasures
You'll collect a digital Treasure from one of these collections when you find and log this geocache:

Loading Treasures