The following earthcache will take you to the Franklin Mineral Museum, home to one of the world's finest collections of UV-reactive fluorescent minerals. By the time you complete this earthcache, you will hopefully know a little more about one of of Northern Jersey's best geological treasures!
Fluoresence vs. Phosphoresence
Fluorescence in minerals occurs when a specimen is illuminated under a specific wavelength of light.
Ultraviolet (UV) light, x-rays, and cathode rays are the typical types of light that trigger fluorescence. These types of light have the ability to excite, or activate, susceptible electrons within the atomic structure of the mineral. These excited electrons temporarily jump up to a higher orbital within the mineral's atomic structure. When those electrons fall back down to their original orbital, a small amount of energy is released in the form of light. This release of light is known as fluorescence. The wavelength of light released from a fluorescent mineral is often distinctly different from the wavelength of the incident light. This produces a visible change in the color of the mineral. This "glow" continues as long as the mineral is illuminated with light of the proper wavelength.

The photo below shows a sample of rocks/minerals under two different wavelengths: normal "daylight" and a shortwave UV light. As you can see, what look like ordinary rocks in daylight have quite the transformation under shortwave UV light.

Phosphoresence, on the other hand, occurs even after the mineral is illuminated after being exposed to the specific wavelength. Fluorescence occurs when electrons excited by incoming photons jump up to a higher energy level and remain there for a tiny fraction of a second before falling back to the ground state and emitting fluorescent light. With phosphorescence, the electrons remain in the excited state orbital for a greater amount of time before falling. Minerals with fluorescence stop glowing when the light source is turned off, while minerals with phosphorescence can glow for a brief time after the light source is turned off. Certain samples of fluorite can phosphoresce seconds or even minutes after exposure to UV light. Willemite (a common mineral found throughout Franklin, NJ) is also known to exhibit these same properties for a few seconds after fluorescing.
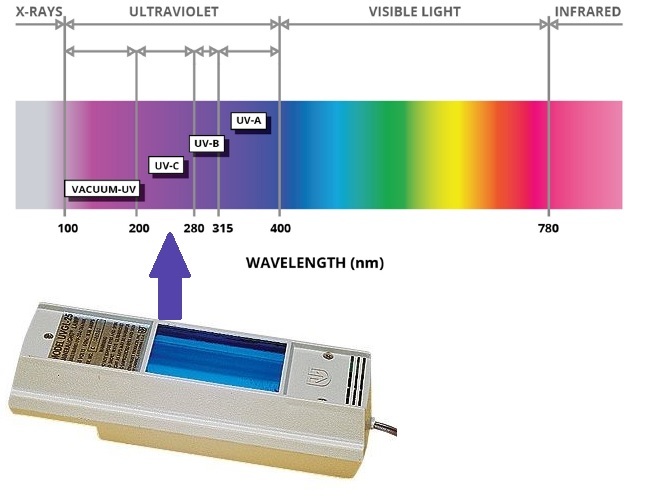
Shortwave vs Longwave UV radiation
One of the best ways to observe fluoresence in minerals is with UV lights. While about 10% of the fluorescent minerals in this area only glow under longwave UV radiation (~365-400nm), the remainder of the fluorescent specimens glow under shortwave UV radiation (~250nm). Unfortunately, shortwave UV lights are more expensive and harder to find. Shortwave UV radiation can also be damaging to your eyes and skin, so be careful with exposure!
What's so special about Franklin/Ogdensburg?
Northern New Jersey is home to some of the best collections of fluorescent minerals in the world. Franklin and Ogdensburg both have rich histories of zinc (Zn) mining. The high concentration of manganese (Mn) within the zinc ores acts as an effective "activator" for the fluorescence in many of the different types of minerals found here. Academic geologists and collectors alike seek out specimens from areas close to this earthcache. There is, perhaps, no better place to go on a fluorescent field trip than this museum and the Sterling Hill Mine in neighboring Ogdensburg! Here are some of the more common minerals that can be found in this area and how they appear under shortwave UV light:
Calcite: fluoresces -red- in color
Clinohedrite: fluoresces -bright orange- in color
Franklinite: does not fluoresce; usually appears -black- in color alongside other fluorescent minerals
Hardystonite: fluoresces -blue- to -violet- in color
Margarosanite: fluoresces -bright blue- in color
Willemite: fluoresces -bright green- in color

A Sterling Hill Mine specimen, under UV shortwave light, with deposits of willemite (green), clinohedrite (orange), hardystonite (blue), and franklinite (black)

Another Sterling Hill Mine specimen. The left photo shows the specimen under "normal" light. Photo on right is under UV shortwave light, showing deposits of calcite (red), franklinite (black), and willemite (green).

Margarosanite, mined from Franklin, under a shortwave UV light.
Touring the Museum:
In order to complete this earthcache (and truly enjoy the scientific and aesthetic wonders of this geologic phenomenon), you will need to visit the museum during its operating hours. Information about prices and times can be found here. The museum is open 11am-4pm on weekdays from April to November and 11am-5pm on weekends from March to November. Unfortunately, the museum is closed entirely from December to February. While we encourage you to visit the entirety of the museum, you will need to find the fluorescent mineral exhibit to complete the logging tasks. (See below....)
Logging Requirements:
Please email us or use the messaging service to submit answers to the following questons:
1.) What about Franklin/Ogdensburg makes it such a good location to find fluorescent minerals?
2.) While touring the fluorescent mineral exhibit, please note what the most prevalent color is and what mineral you hypothesize it to be (given the short list above).
3.) With your back to the larger specimens, look at the various display cases along the wall. What makes the one on the far left different from the rest?
4.) Turn off the UV light and examine the rocks carefully. What evidence, if any, did you see of phosphoresence?
5.) How might fluorescence be a useful identification tool for geologists/mineralologists?
6.) OPTIONAL: Post some photos of your tour.
Logs that do not meet the requirements for #1-5 above (via email or message) will be deleted!
Works Cited in this Earthcache:
Bostwick, R. C. (2015). A 2015 Checklist Based on Observations of Confirmed Specimens. Retrieved from https://franklinmineralmuseum.com/about/fluorescent-minerals/
King, H. (2005). Fluorescent Minerals. Retrieved from http://geology.com/articles/fluorescent-minerals/
Warren, T. S., Gleason, S., & Bostwick, R. C. (1999). Ultraviolet Light and Fluorescent Minerals: Understanding, Collecting and Displaying Fluorescent Minerals. Baldwin Park, CA: Gem Guides Book Co.
A special thanks is in order to the Franklin Mineral Museum for allowing the placement of this earthcache and providing a lot of valuable educational information about their specimens. You guys rock! (pun-intended)