Ah yes, that dreaded warning that you didn't notice, until you got home...
BATTERIES NOT INCLUDED!

Go figure - I went out and hid a cache around here somewhere and - coordinates not included!
The cache will be found at:
N047 34.ABC W052 41.DEF
where A through F are determined by looking into the following factoids concerning battery history and battery trivia.
For A: Allesandro Volta was the first to produce electricity in a battery-type sense with his "voltaic pile":

Volta stacked several pairs of alternating copper and zinc discs (lectrodes) separated by cloth or cardboard soaked in brine. When the top and bottom contacts were connected by a wire, an electric current flowed through the voltaic pile and the connecting wire. Volta, whom the word "voltage" for electrical potential was coined, lived from 174X – 18Y7. A = X - Y.
For B: If you study introductory electrochemistry - the study of how chemical processes can be used to produce or store electric potential - you will likely come across the Daniell Cell fairly early into your studies. Like the Voltaic Pile, the Daniell Cell produces a voltage from a chemical reaction, yet it packaged the reaction in a more enclosed, less corrosive to surroundings setup.
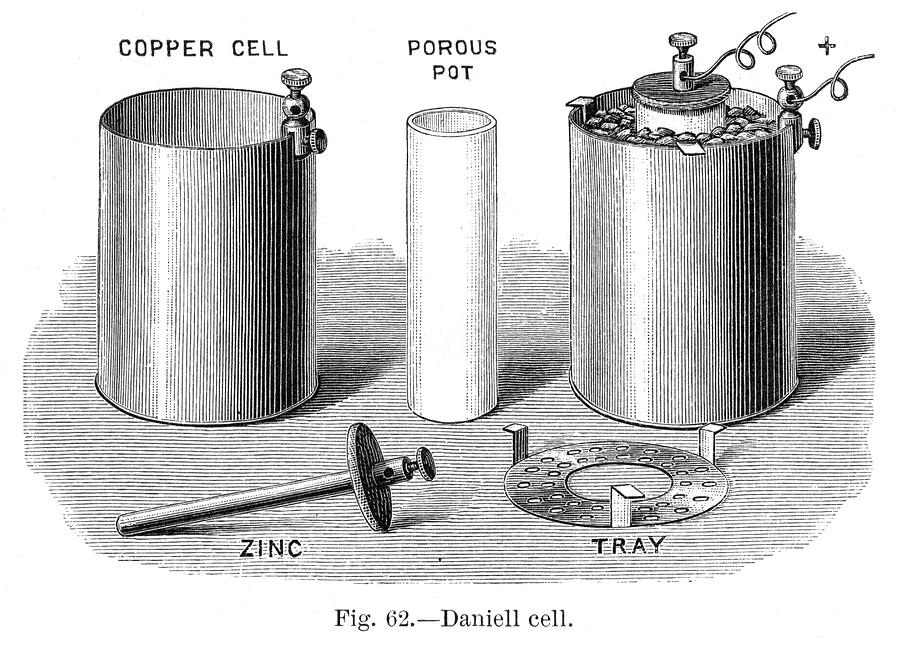
The key concept to the Daniell Cell is the porus cup - a clay pot which was not glazed, allowing to have ions migrate through the copper side and the zinc side of the cell. This allows the electrochemical reaction to occur in, once assembled, a totally enclosed system, producing 1.1V per cell. This electrochemical cell design was designed in 18B6 by John Frederic Daniell, a chemist and meteorologist.
For C: Gaston Planté invented the lead–acid battery, the first-ever battery that could be recharged by passing a reverse current through it. This type battery was developed as the first rechargeable electric battery marketed for commercial use and it is the same concept widely used in automobiles today.

His invention was in the year 18C9.
For D: A spring-top lantern battery is a huge commercial battery compared to most, yet is proof that the voltage produced by a battery is not determined by its physical size. These large batteries are larger to have more capacity of chemical reactant inside, to last longer before requiring replacing.

In North America, the typical voltage output of a spring-top lantern battery is DV.
For E: While we may be used to battery size designations such as "AAA" or "C", these designations are not the only size designations, and some of these designations may differ from one country to the next.

The International Electrochemical Commission (IEC) is an organization which publishes codes for battery sizes which are not country nor brand specific. For example, a "D" sized cell battery is also a "RE0" sized battery according to the IEC.
For F: Watch batteries - circular shaped "button batteries" - are notorious for having several codes to mean the same battery. In many cases, manufacturers have their own code for the battery, so when they suggest a replacement, the consumer specifically buys their brand again as they search for the exact same replacement code. A typical button battery is shown in the image below.

Note the bottom of the pic: This battery is the same as AG11, LR721 (stamped on the battery), or 362. A 362 can also be replaced by a 361! So before going out and paying a ton of cash for a specifically coded watch battery, check to see what other codes are equivalent, and likely find an equally effective, cheaper battery for your purposes! A 362 watch battery, according to international IEC codes is a SR5F battery.
So: Do you have your geocaching hunger recharged? Check your coordinates into the checker below, and then off you go to make the find!