Iceland Spar the Gem of Iceland
Introduction
Iceland Geology: Geologically Iceland is still very young - just 20-25 million years old. It lies exactly at the interface of two continental plates. This ensures a high level of volcanic activity right up to the present day.
Deep crevices, trenches and canyons bear witness to the tectonic, volcanic and erosive forces that still shape the Icelandic landscape today.
There are four major geological forces that have shaped Iceland over millions of years: plate tectonics, volcanism, ice ages and erosion.
Origin and distribution of Island Spar
The formation of Iceland spar is closely related to the formation of magmatic rocks. Hydrothermal solutions circulating in these rocks crystallize as isspath when cooled in corridors or rock cavities. However, the mineral can also be contained in rocks as a mixed component. Furthermore, isspath is also found in carbonate-rich sedimentary rocks.
Calcite is widely distributed as a secondary mineral in the basaltic lava sequences of Iceland, with crystals generally less than a few centimeters in size. However, there are few exceptional locations in Iceland where large calcite crystals have been found. The best-known locality is 1 km east of the farm Helgustadir within the central volcano Reydarfjördur. Probably they were deposited over a long period due to some special local conditions prevailing in the groundwater circulation. Observers' reports vary, but the calcite crystals apparently appeared in veins and bends in a subvertical zone with dimensions in the order of 4x15 m through the lava flows. Single crystals could reach a size of three meters or even more. The best specimens were found where the lavas had been completely transformed into clay.

A preserved mine with an interesting history
In Iceland - a mountain full of the material Spar. It lay in a remote place near the farm Helgustaðir. It simply existed to please those who lived in the area or traveled by.
In a basalt cavern at Helgustaðir farm, large, clear crystals were found for the first time and enabled the physician Erasmus Bartolinus to study birefringence from 1669.
Then Iceland Spar became a commodity and mining began.
But its history goes back far before that time. It is believed that the Vikings used this mineral as a navigator because of its light polarizing ability, so they could always find the sun in any weather. Historians believe that the "sun stone", in reality Iceland Spar.
This deposit of outstanding transparent Calcite was discovered in the 17th century and was intermittently mined for many decades until 1924.
The transparency of the crystals were ideally suited for the optical industry and therefore the Calcite quickly became famous as "Iceland Spar" and its marvelous crystals were rapidly distributed among the leading European scientists of that time.
Icelandic Spar has long been used in optical instruments (e.g. polarisation microscopes). The double spar refracts a light beam into two different beams, resulting in a double contour of the observed object.
The initial concepts of double refraction and crystal cleavage planes were first discovered on Calcite crystals from this site and led to a far better understanding of crystal physics.
Hence Helgustadir is one of those classic sites, which started mineralogy as a serious science.
As a result, large quantities were exported to Europe from the 17th century until the quarry was closed in 1924.
Today Helgustaðir is preserved. It is partly open for visitors. Inside the cave, left over from days of mining, you can still see rocks sparkling with Calcite.
The area is a nature reserve since 1975.
It is strictly forbidden to take even the smallest samples from the quarry.
"Viking sun-stone”
Today Iceland Spar is primarily of interest to collectors. Historically, however, it has been used both in navigation and in science.
Iceland Spar has a unique property: A ray of unpolarized light, like sunlight, passing through the crystal divides into two rays of perpendicular polarization directed at different angles. This double refraction can be used to find the precise location of the sun, even when the sun is completely hidden by clouds or the horizon during twilight.
Calcite (Iceland Spar)
The most important characteristics of the Icelandic Spar in Helgustaðir are size, purity and clarity.
Iceland Spar is especially transparent Calcite. Clear, pure Calcite crystals act as optical crystals, but mostly they are fission Rhombohedra.
A special feature of crystalline Calcite is the strong birefringence of the light: light that is not incident along the optical axis of the crystal is split into two light bundles, one ordinary and one extraordinary. These two beams have a different refractive index due to different polarization directions. This is shown by the fact that at a certain angle of view, every object observed through a clear crystal appears double, hence the name double spar (Iceland Spar).

Features
Colour clear-colourless Hardness 3
Transparency transparent Bruch conchoidal, brittle
Mineral class Carbonates Fissionability perfect
Formula CaCO3 Gloss glass, matt, silk gloss
Cristallsystem trigonal Density 2,6 – 2,8
Calcite crystals are trigonal-rhombohedral. They show a remarkable variety of habits, among them pointed to blunt Rhombohedra, tabular forms, prisms or different Scalenohedra. Calcite has several twin species, which further increases the variety of forms observed. It can be fibrous, granular, lamellar or compact. It usually splits in three directions parallel to the Rhombohedron shape.
Calcite is the mineral in nature with the widest range of forms, accounting for a mass proportion of approx. 4 percent of the earth's crust.
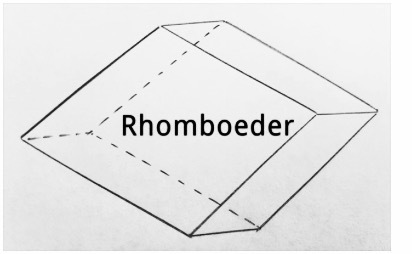
The Rhombohedron can be found in nature as a crystal form and on an atomic level in crystal structures. It is limited by 6 rhombuses. When breaking Calcites, the fragments always split in the form of the Rhombohedron.
Rhombohedra are the most frequently observed Calcite forms. The steepness of the faces and the angles can differ.
The mineral with the most forms on Earth no other mineral shows so many different crystal forms and combinations. Actually, with only a few basic forms Calcite has developed a crystalline magic world, which amazes collectors and crystallographers worldwide. The pseudomorphoses and the unique Calcite twins are especially sought after. The birth of the modern mineralogy Calcite shows some physical properties so extraordinarily clearly, that it became almost a symbol for it: fissility, fluorescence and birefringence. 200 years ago, fissility was even the cause of important disputes among scientists. It is even said that the solution to this problem was the birth of modern mineralogy.
Please answer the following questions:
Information board
Here you can find out about the history of mining here.
Follow the small path up to the entrance of the mine:
N 65° 02.000 W 013° 51.225
Find a larger calcite rhombohedron at the entrance of the mine.
- Describe the different layers of rock at the entrance of the gallery, what conclusion you draw in connection with the formation of Iceland spar.
- Now try to describe the rhombohedron you found in your own words. (colour(s), geometrical shapes, structure, size)
- What is the habit (shape) of these rhombohedra? (Do they grow straight, radially or in any direction?)
- Try to scratch the surface of a crystal slightly with a coin or stone. What could this tell us about the hardness of calcite?
- Birefringence: Voluntary experiment:
Put your clear Iceland Spar on a paper on which you have drawn a cross.
Now look at the cross vertically through the Iceland Spar. Now slowly turn the Iceland Spar around its own axis.
Observation: What happens with the cross?
- Photos - as a supplement to the tasks are again allowed and expected from us as log condition. Please take a photo of your rhombohedra with your CACHERNAMES on a piece of paper or of yourself.
After sending your answers you can log in immediately, if something is wrong, we will contact you.
The correct answers with your photo from the site will then be considered as log-in authorisation!
You do not need to wait for our log release!
We wish you lots of fun on this geological expedition!
If you enjoyed your trip, we would of course be pleased to receive many photos in the log.
Team sissifalke
ATTENTION!
It is strictly forbidden to take even the smallest samples from the quarry.
Sources:
https://notendur.hi.is/leo/pdf/Iceland%20Spar%20Brochure-Corr2.pdf
https://de.wikipedia.org/wiki/Calcit