This is a radioactive synthetic element whose most stable isotope is 268Db with a half life of 28 hours. This is the longest lived transactinide isotope and is a reflection of the stability of the Z = 108 and N = 162 closed shells and the effect of odd particles in nuclear decay. Chemistry experiments have provided sufficient evidence to confidently place dubnium in group 5 of the Periodic Table.
Discovery profile
Element 105 was first reported by Russian scientists in 1968-1970 at the Joint Institute for Nuclear Research in Dubna, Russia. The 1968 work was based on the detection of correlated decays of element 105 to known daughter nuclei using the reaction 243Am(22Ne,xn). They reported a 9.40 MeV and a 9.70 MeV alpha-activity and assigned the decays to the isotopes 260105 or 261105.
Proposed names
Historically element 105 has been called eka-tantalum, reflecting Mendeleev's placeholder terminology.
The American team proposed that the new element should be named hahnium (Ha), in honor of the late German chemist Otto Hahn. Consequently this was the name that most American and Western European scientists used and appears in many papers published at the time.
The Russian team proposed the name nielsbohrium (Ns) in honor of the Danish nuclear physicist Niels Bohr.
An element naming controversy erupted between the two groups. The International Union of Pure and Applied Chemistry (IUPAC) thus adopted unnilpentium (Unp) as a temporary, systematic element name. Attempting to resolve the issue, in 1994, the IUPAC proposed the name joliotium (Jl), after the French physicist Frédéric Joliot-Curie. The two principal claimants still disagreed about the names of elements 104-106. However in 1997 they resolved the dispute and adopted the current name, dubnium (Db), after the Russian town of Dubna, the location of the Joint Institute for Nuclear Research. It was argued by IUPAC that the Berkeley laboratory had already been recognized several times in the naming of elements (i.e., berkelium, californium, americium) and that the acceptance of the names rutherfordium and seaborgium for elements 104 and 106 should be offset by recognizing the Russian team's contributions to the discovery of elements 104, 105 and 106.
Spectroscopic decay level schemes for dubnium isotopes 257Db
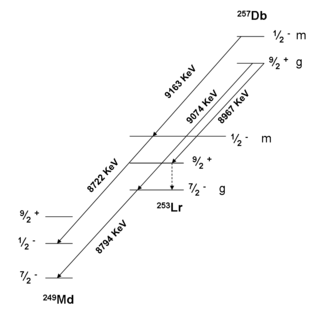
This is the currently suggested decay level scheme for 257Dbg,m from the study performed in 2001 by Hessberger et al. at GSI
Experimental chemistry
Gas phase chemistry
The chemistry of dubnium has been studied for several years using gas thermochromatography. The experiments have studied the relative adsorption characteristics of isotopes of niobium, tantalum and dubnium radioisotopes. The results have indicated the formation of typical group 5 halides and oxyhalides, namely DbCl5, DbBr5, DbOCl3 and DbOBr3. Reports on these early experiments usually refer to dubnium as hahnium.
PERIODIC TABLE

Check out this interactive Periodic Table.
Check out this Dubnium video. Prepared by The University of Nottingham.